What Is the Basic Principle of Thin Layer Chromatography
The compounds in the mobile phase move over the surface of the stationary phase. Thin Layer Chromatography Principle.

Thin Layer Chromatography Tlc Principle Procedure Applications On Byju S
Similar to other chromatographic methods thin layer chromatography is also based on the principle of separation.

. In order to have paper chromatography with other stationary phases such as silica alumina etc a thin layer chromatography was invented. First week only 499. This layer of adsorbent is known as the stationary phase.
In chromatography a mixture dissolves in a solvent known as the mobile phase and is carried up a solid known as the stationary phase. The process begins here by moving the mobile phase over the stationary phases surface. Thin Layer Chromatography Principle.
Principles and practice of thin layer chromatography Thin layer chromatography TLC is an invaluable method used in chemistry and biochemistry for the separation and analysis of a wide variety of molecular mixtures. The high-performance thin-layer chromatography is also known as flat-bed chromatography or as planar chromatography. The distinction depends on the relative affinity between the stationary and mobile phases of the compounds.
Silica is also acidic. Apr 2 2020 HPLC Basics. Basic Principle of Thin Layer Chromatography In Thin layer chromatography TLC the analytes or sample mixture is applied as a minute dot or spot at the bottom of the stationary phase it holds up on a glass metal or plastic plate.
Development consists of placing the bottom of the TLC plate into a shallow pool of a development solvent which then travels up. Thin-layer chromatography also known as TLC is a separation technique used to separate and analyse components in a mixture. One mobile phase and one contiguous stationery phase.
Solution for What is the basic principle for Thin layer chromatography. A solvent of the sample is allowed to evaporate from the plate which is after that positioned in a closed chamber in which at the bottom there is a. Paper chromatography can also be used for this purpose.
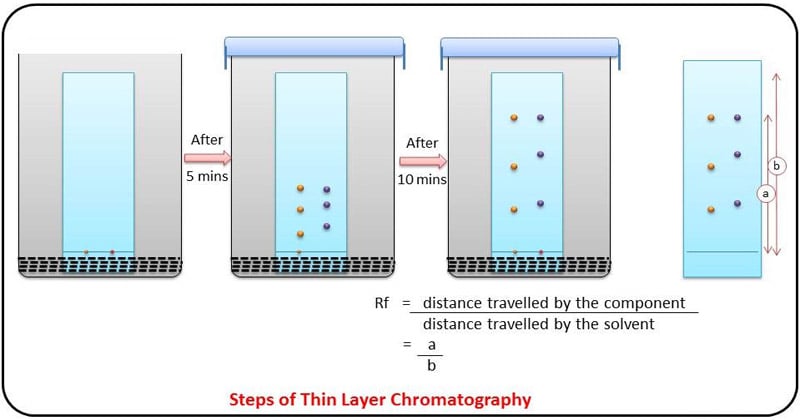
The compounds under the influence of the mobile phase driven by capillary action travel over the surface of the stationary phase. In this method silica gel is act as a stationary phase and natural sources to be studied act as a mobile phase. Separated components are identified based on R f value.
High-Performance Thin-Layer Chromatography HPTLC Principle The HPTLC works on the same principles as TLC such as the principle of separation is adsorption. Start your trial now. We will begin with the TLC principle.
Based on the physicochemical characteristics of the analyte and the availability of instrumentation a chromatographic system is chosen to separate identify and quantify the analyte. Thin-layer chromatography Thin-layer chromatography is a solid-liquid adsorption chromatography. Paper chromatography was well known in the 1930s it was relatively faster but you are limited by choice of stationary phases just cellulose.
Principles of Thin layer chromatography Thin layer Chromatography consists of two phases. Both of these adsorbents are polar but alumina is more so. In this method stationary phase is a solid adsorbent substance coated on glass plates.
Thin Layer Chromatography TLC is a solid-liquid technique in which the two phases are a solid stationary phase and a liquid moving phase. In this case non polar compounds are more soluble higher Rf values and polar compounds are more adsorbent lower Rf values. Thin-layer chromatography is a method of separation or identification of a mixture of components by using finely divided adsorbent solid liquid over a glass plate and liquid as a mobile phase.
The mobile phase or solvent flows through the capillary action. Thin Layer Chromatography Principle The separation principle of the TLC procedure is based on the given compounds relative affinity towards the mobile and the stationary phase. This chapter gives the basics of chromatography as a separation method based on the partitioning of a solute between a mobile phase and a stationary phase.
What is the principle behind the thin layer chromatography TLC of lipids. Thin layer chromatography TLC is a type of chromatography where the stationary phase is a glass plate coated in the absorbent material often silica gel or alumina and the mobile phase is an organic solvent. The stationery phase is a thin layer of silica or alumina coated on glass plastic or.
The components within the mixture. To study this thin layer chromatography TLC is a simple and efficient technique. The plate used for this process is known as chrome plate.
TLC is a separation technique where the molecules of mixtures separate using differential migration through a stationary phase the solvent mixture flowing through the virtue of capillary forces. Principle of Thin Layer Chromatography TLC Thin-layer chromatography is performed on a sheet of glass plastic or aluminium foil which is coated with a thin layer of adsorbent material usually silica gel aluminium oxide alumina or cellulose. Thin Layer Chromatography Principle.
In column chromatography alumina silica gel cellulose can be utilized. Thin layer chromatography is based on the separation of a mixture of compounds as it migrates with the help of a suitable solvent through a thin layer of adsorbent material which has been applied to an appropriate support. Thin Layer Chromatography In the process of thin-layer chromatography TLC the mixture of substances is separated into its components with the help of a glass plate coated with a very thin layer of adsorbent such as silica gel and alumina as shown in the figure below.
The separation relies on the relative affinity of compounds towards both the phases. The separation depends on the relative affinity of compounds towards the stationary and the mobile phase. As adsorbent material all solid substances used.
Solids most commonly used in chromatography are silica gel SiO 2 x H 2 O and alumina Al 2 O 3 x H 2 O. Weve got the study and writing resources you need for. In Thin layer chromatography TLC the analytes or sample mixture is applied as a minute dot or spot at the bottom of the stationary phase it holds up on a glass metal or plastic plate.
Like other chromatographic techniques thin-layer chromatography TLC depends on the separation principle. Thin layer chromatography or TLC is a method for analyzing mixtures by separating the compounds in the mixture. Thin-Layer Chromatography - Key takeaways.
Thin Layer Chromatography Definition Principle Parts Steps Uses

Comments
Post a Comment